Flow Cytometry Services
State-of-the-art high-complexity flow cytometry throughout all stages of drug development
- Analytical Assays For Endpoint Data Collection
- Flow Cytometry Services
At Champions Oncology, we offer an extensive range of flow cytometry services tailored to support your research. Our capabilities include clinical flow cytometry, which ensures robust and accurate data in clinical trials. From custom flow panel design to fit-for-purpose validation, we provide specialized assays that deliver deep insights into immune response and tumor interaction. By leveraging both conventional and spectral cytometry technology, we help streamline complex flow cytometry studies in oncology, immuno-oncology, and hematology applications.
.png?width=1176&height=614&name=MicrosoftTeams-image%20(41).png)
Industry Leader in Complex Flow Cytometry Solutions
Flow Cytometry is a powerful analytical tool to evaluate, assess, and quantify complex responses and critical phenotypical changes. With significant advances in the development of drugs modulating the immune system, Champions' preclinical and clinical flow cytometry services are implemented using fully optimized, validated, and GCLP-compliant panels, developed to be efficiently customizable to meet your needs:
-
Champions offers full custom development of multi-parameter panels, suitable for multi-site harmonization to support global clinical trials
-
Industry best timelines featuring expedited development and optimization to meet your milestones
-
Operational expertise with quality assurance and GCLP regulatory compliance for preclinical and clinical research
-
Ideally positioned to provide meaningful insights for your preclinical studies and support exploratory and secondary endpoints for global clinical trials
Advanced Preclinical Flow Cytometry with Custom Panel Development
Champions uses flow cytometry in most preclinical oncology studies to characterize tumors and quantitatively measure relevant phenotypical changes associated with drug responses in immune-competent models including syngeneic and humanized mice and ex-vivo co-culture systems.
We provide our off-the-shelf panels or custom flow cytometry panel design. We can also transfer your in-house developed panels to be executed in our labs at Champions, saving you time and resources.
Champions has expertise in several sample types as well as experience working with different species, such as mice, rats, rabbits, non-human primate (NHP), and human samples.
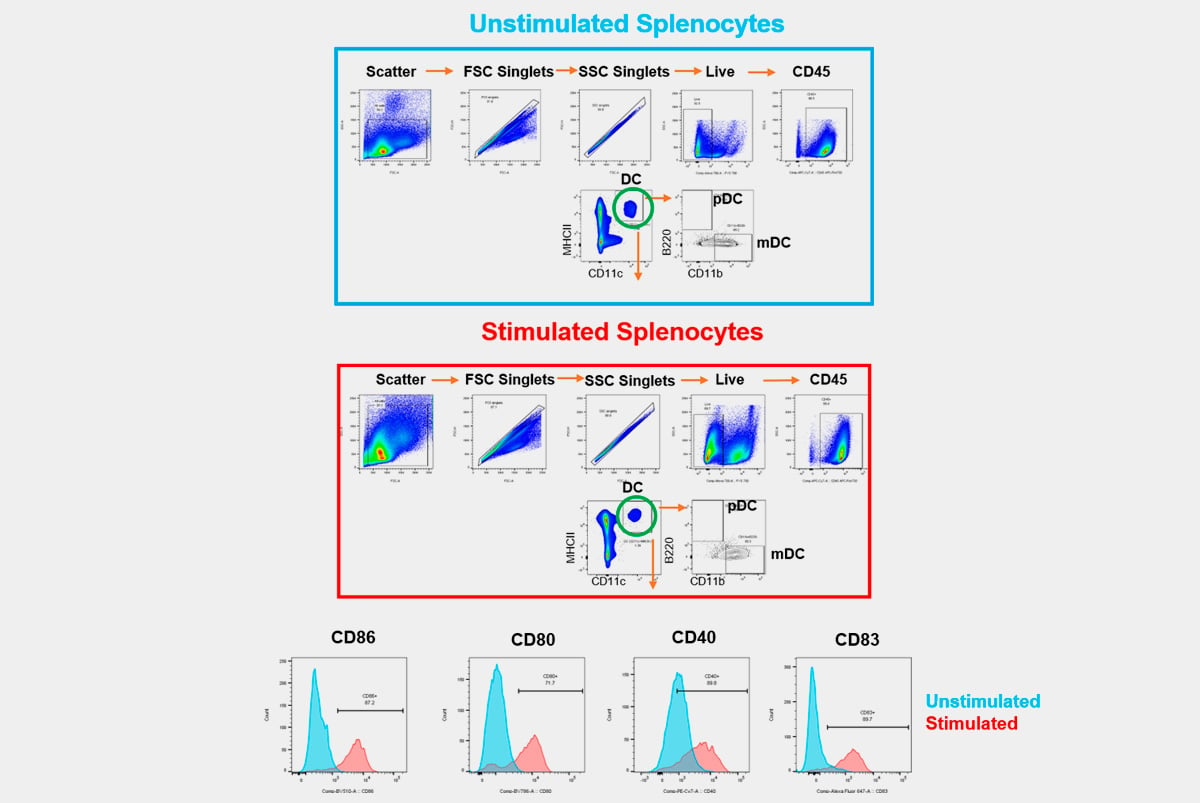
High-Complexity Clinical Flow Cytometry for Specialty Testing
Champions' GCLP-compliant clinical flow cytometry services are implemented using fully optimized and validated human panels developed to meet your clinical trial needs.
We understand that every flow cytometry assay is unique, and therefore, we offer custom assay development and fit-for-purpose validation to suit our clients' needs. Champions is a leader in high dimensional flow cytometry and knows how important receiving high-quality, reproducible data is to you – we do our best to do it right, with scientific integrity.
Champions leads the industry in clinical flow cytometry expertise and innovative technology and will provide you with the highest quality, complex computational results with the ability to interrogate up to 30 parameters on each individual cell simultaneously within every patient sample. We also have experience executing challenging flow cytometry-based receptor occupancy (RO) assays, intracellular cytokine assays (ICS), and phosphorylation-based functional assays.
"We are thoroughly impressed with Champions Oncology’s flow cytometry services. Their experts have delivered clear, actionable results that have significantly accelerated our research progress."
___________________________________________________
"Their precise cell analysis has been crucial for our Clinical Trials, offering valuable data that has advanced our understanding of immune cell functions and therapeutic responses."
___________________________________________________
"Their custom analysis and high-quality results have greatly enhanced our ability to study complex cell populations and validate our therapeutic targets."
___________________________________________________
In vivo models for numerous diseases and conditions have endpoints that have involved animals being falling ill or dying. As researchers, we have sought to use animal models in more humane and practical ways using surrogate endpoints that have been developed to prevent animals from suffering while still providing critical research data. Flow cytometry has been instrumental in these advances.
Immunotherapies are focused on strategies that alter immune responses, using antibodies that bind to receptors on different immune cell subsets to activate or suppress immune cell function. As such, it was necessary to develop assays that assess the functional and biological effect of a therapeutic on its target. When incorporated into high-parameter flow cytometry panels, receptor occupancy assay can simultaneously evaluate receptor expression and drug occupancy on defined cell subsets, to provide functional effects and safety information.
Ready to start working with Champions Oncology?
Accelerate your research with cutting-edge flow cytometry services from our expert team. Whether you require a custom flow panel or a highly specific T cell activation flow cytometry assays, Champions Oncology is equipped with the latest technologies and expertise to provide tailored, high-quality results. Reach out now to discuss how we can assist in advancing your research goals.
What is flow cytometry used for in oncology?
Flow cytometry is a powerful analytical tool used in oncology to measure and analyze multiple phenotypic characteristics of cells as they flow in a fluid stream through a laser. It is widely used in oncology for diagnosing and classifying cancers, particularly hematological malignancies like leukemia and lymphoma. Flow cytometry helps identify specific cell populations based on surface markers, assess cell proliferation, determine apoptosis and cell death, and detect minimal residual disease (MRD). It is also instrumental in evaluating the immune profile of tumors and monitoring disease progression or response to therapy.
How can flow cytometry services support in vivo models?
Flow cytometry services can greatly enhance in vivo models by providing detailed, quantitative analysis of cell populations within tissues or blood samples. These services enable the assessment of immune cell infiltration in tumors, characterization of circulating tumor cells, and monitoring of immune responses in preclinical models. By analyzing cells from in vivo models, flow cytometry can offer insights into the efficacy of therapeutic interventions, such as the activation or suppression of specific immune cell types, thus aiding in the development of cancer therapies.
What is the typical workflow for a flow cytometry experiment?
The typical workflow for a flow cytometry experiment involves several key steps:
- Sample Preparation: Cells are harvested, counted, and often labeled with fluorescently conjugated antibodies that target specific markers.
- Fixing: Cells are fixed with formaldehyde in order to preserve the structure of the cells as well as keep markers intact throughout the experiment.
- Permeabilization: It is required to permeabilize cells prior to staining for the detection of intracellular cytokines.
- Blocking: Depending on the experiment and marker specificity, it is important to block for Fc receptors to prevent additional background stain uptake to the cells. If you are determining intracellular cytokine production for a particular cellular subset, it is important to block the secretion of cytokines with Brefeldin A and/or Monensin, which results in the increased accumulation of cytokines in the cell.
- Staining: The prepared cells are incubated with the chosen antibodies or dyes. Specific negative and positive controls must be created to ensure experimental analysis is completed correctly.
- Data Acquisition: The stained cells are passed through the flow cytometer, where lasers excite the fluorophores, and detectors measure the emitted light, quantifying various cell properties.
- Data Analysis: The collected data are processed using specialized software to interpret the results, such as identifying cell populations, analyzing marker expression, or assessing cell viability.
How does flow cytometry help in monitoring treatment response in cancer?
Flow cytometry plays a crucial role in monitoring treatment response in cancer by allowing for the detailed analysis of how cancer cells and the immune system respond to therapy. It can detect changes in cell populations, such as the reduction of cancer cells or the activation of immune cells, in response to treatments like chemotherapy, immunotherapy, or targeted therapies. Flow cytometry also enables the detection of minimal residual disease (MRD), helping to assess whether a treatment is effectively eliminating cancer cells or if there is a need for alternative therapeutic strategies.
How is flow cytometry used in immuno-oncology research?
Flow cytometry is extensively used in immuno-oncology research to analyze the immune landscape within tumors and the systemic immune response to cancer. It allows researchers to identify and quantify different immune cell populations, within the tumor microenvironment or peripheral blood. Flow cytometry is also used to assess the expression of immune checkpoint molecules, cytokines, and other markers that indicate immune activation or suppression. This information is critical for understanding how tumors evade the immune system, mechanisms of resistance, and for developing and optimizing immunotherapies.